Not yet available in the U.S. or Canada
A Fast, Simple, Capillary Blood Analyzer
• Capillary fingerstick samples for all blood tests
• Immediate test results during the patient visit
• Reduces patient follow-up visits and costs
Allegro is a compact, point-of-care (POC) analyzer that features a clinically important menu of 10 measured and individually selectable tests, plus 7 calculated tests. All tests are measured with disposable, ready-to-use cartridges or test strips, and are easily performed by non-technical personnel.
Allegro uses a very small capillary blood drop for all blood tests, which eliminates the need for venipunctures or a trained phlebotomist. With cartridge test times of 6 to 9 minutes and the ability to measure two cartridges simultaneously, Allegro provides rapid results. Having results during the patient visit—not after—is more effective for patient/clinician meetings; reduces office calls, letters, and follow-up visits; and improves patient compliance with care recommendations.
A comprehensive test menu for monitoring glycemic control and diabetes risk factors
Allegro provides a comprehensive test menu to monitor the effectiveness of glycemic control, assess the risk or progression of diabetes complications, and make therapeutic adjustments for patients.
| Glycemic Control | Kidney Function | Lipids/Cardiac Risk | Coagulation | Inflammation |
| HbA1c | Blood Creatinine | Total Cholesterol | PT/INR | CRP |
| Glucose |
eGFR | HDL Cholesterol | ||
| Estimated avg. Glu | Urine Creatinine | Cholesterol/HDL Ratio | ||
| Urine Albumin | Non-HDL Cholesterol | |||
| Albumin/Creatinine Ratio | LDL Cholesterol | |||
| Triglycerides |
The only analyzer that measures all blood tests from a capillary sample
Capillary sampling is easily performed by staff members, eliminating the need for venipunctures, trained phlebotomists,
and blood drawing tubes and needles. Allegro’s use of a small,
5 μL blood drop makes testing virtually painless for the patient.
Tests Available from Capillary Fingerstick Samples
| Test | Allegro | DCA Vantage | Afinion |
| HbA1c | ✓ | ✓ | ✓ |
| Lipids Panel |
✓ | ✓ | |
| Blood Glucose | ✓ | ||
| Blood Creatinine | ✓ | ||
| PT/INR | ✓ | ||
| CRP | ✓ | ✓ |
Tests Available from Urine Samples
| Test | Allegro | DCA Vantage | Afinion |
| Urine Creatinine | ✓ | ✓ | ✓ |
| Urine Albumin |
✓ | ✓ | ✓ |
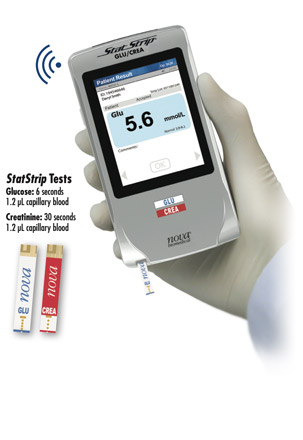
Capillary Sample Meter Tests
The StatStrip meter wirelessly transmits patient-identified test results to the analyzer where they are combined with the cartridge test results in a single patient report.
• Lab-like results
• The only glucose meter FDA
cleared for critical care
Capillary Sample Cartridge Tests


HbA1c
6.9 minute assay
1.5 μL capillary blood

Lipids
10.4 minute assay
5.0 μL capillary blood
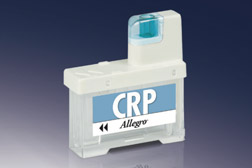
CRP
7 minute assay
5.0 μL capillary blood

PT/INR
< 8 minutes assay
5.0 μL capillary blood

Urine albumin/creatinine
7.5 minute assay
25 μL urine
Easy-to-use Analyzer
Simple Testing Procedure
Allegro’s simple testing procedures are easily performed by office staff. An onboard barcode scanner automatically enters patient ID and cartridge test assay information. When an analysis is completed, the results are automatically printed and stored in the Allegro database and transmitted to the electronic medical record (EMR) for clinician viewing.
Cartridge Testing

Scan cartridge
Perform fingerstick

Remove capillary sampler

Touch sampler to blood

Replace capillary sampler

Insert cartridge and press
"Analyze"
Meter Testing

Insert test strip
Perform fingerstick
Touch strip to blood
For more product information, click here or contact Nova Biomedical at:
Nova Biomedical / 200 Prospect Street / Waltham, MA 02454 / 781-894-0800
For Nova Biomedical Sales inquiries, reach us at 1-800-458-5813.
For Technical Support, reach us at 1-800-545-NOVA (6682).
To place an order, call 1-800-822-0911 or email novaorders@novabio.com
Compact size for POC use
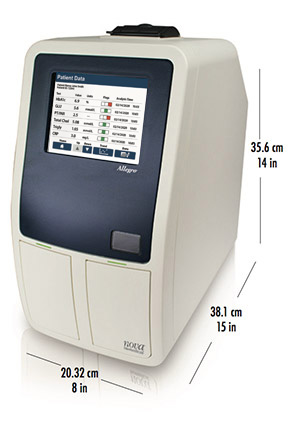
Allegro’s 20.32 cm x 38.1 cm footprint easily fits in clinician offices, clinics, and hospital outpatient locations. It can also be operated on an optional mobile cart.
Physical Specifications:
Width: 20.32cm (8in)
Height: 35.6cm (14in)
Depth: 38.1cm (15in)
Weight: 10.43kg (23lb)
Electrical Power Requirements:
90-264 VAC, 50/60 Hz
Interfaces:
ASTM Protocol, via serial RS232 TCP/IP, POCT1-A2, HL7
Printer:
Built-in thermal
FDA Labeling:
For in-vitro diagnostic use, pending FDA clearance
CE Marked
StatStrip Meter Specifications
Calculated Tests:
Estimated glomerular filtration rate
Physical Specifications:
Weight: 220 g (0.49 lb)
Size: 147 mm x 79 mm x 30 mm
(5.8 in x 3.1 in x 1.18 in)
Meter Data Storage:
| Patient Tests | 1,000 Tests |
| QC Tests | 200 Tests |
| Users | 4,000 Users |
Connectivity:
Data Output: RJ-45 Ethernet Port
Protocol: TCP/IP Ethernet 100 Mbit
Standard: POCT1-A2 Compliant