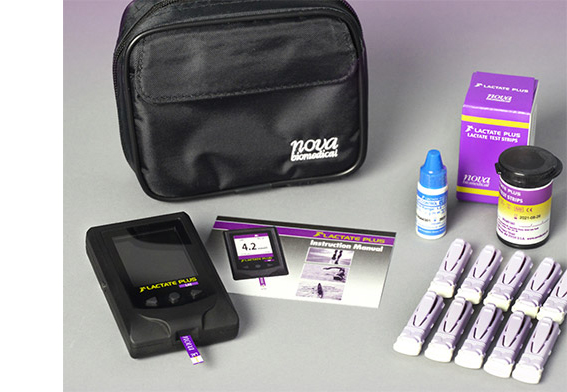
 Lactate Plus Meter
Lactate Plus Meter
Blood Lactate Meter Optimizes Endurance Training and Conditioning
Fast, Virtually Painless Lactate Testing
Purchase here
The Test
Lactate Plus measures blood lactate, a muscle performance indicator, very quickly with a small drop of blood. This provides a virtually painless, yet fast and accurate test for lactate. Blood lactate is the premier indicator of aerobic conditioning for athletes and coaches and a key predictor of performance.
Fast Analysis Time
Lactate Plus provides results in only 13 seconds. This enables athletes to quickly assess performance and adjust training strategies.
Lab-Like Accuracy
Lactate Plus is made by Nova Biomedical, the global leader in hospital- based blood lactate analyzers. More hospitals worldwide use Nova analyzers for lactate measurement than any other company. The same high standards for accuracy and reliability that are required for hospital use have been incorporated into the Lactate Plus meter.
Fast, Simple Testing



Less Pain
Lactate tests on other meters are painful and therefore difficult to incorporate into routine training. They need 5 to 25 microliters of blood and require a wide-gauge lance to pierce the skin deeply enough to obtain a large sample. Lactate Plus requires only 0.7 microliters of capillary blood. This allows for the use of the thinnest, most shallow, and least painful lancing devices available, which increases the athlete’s tolerance for testing.
Large Color Display
Lactate Plus features a large color display that’s easy to read even when outdoors. 
Ergonomic Non-slip Design
The ergonomic non-slip case ensures a secure and comfortable grip for testing.
Test Strip Ejector
An integrated test strip ejector improves operator safety when testing multiple athletes.
Eliminates Calibration Codes
Most lactate test strips require a unique calibration code to be entered manually or by inserting a coding strip before testing with each new lot of test strips. Using the wrong code can change the lactate result by as much as 30%. Lactate Plus strips do not require calibration codes or coding strips, eliminating both a time-consuming step and a source of error.
Low Cost
Lactate Plus meters and strips are priced less than most other similar devices.
Purchase here
For more product information, click here or contact Nova Biomedical at:
Nova Biomedical / 200 Prospect Street / Waltham, MA 02454 / 781-894-0800
For Nova Biomedical Sales inquiries, reach us at 1-800-458-5813.
For Technical Support, reach us at 1-800-545-NOVA (6682).
To place an order, call 1-800-822-0911 or email novaorders@novabio.com
For Sports and Fitness Professionals Only
Precaution: The Lactate Plus System is intended for sports training and conditioning only.
Certifications and Compliance: FDA Quality System Regulations and EN ISO 13485:2016, Complies to IVDD Tested according to: EN 61010-1:2010, EN 61010-2-101:2015, EN 60825-1/A1:2014, UL, CSA Standards. Patented Technology. Patents 6,287,451 / 6,837,976 / EP 1 212 609 / CA 2,375,092 / EP 1497 449 / JP 405 0078 Specifications subject to change without notice.