StatSensor® and StatSensor Xpress™ Creatinine and eGFR Meters

The StatSensor Creatinine Hospital Meter System is a handheld analyzer and miniaturized, single-use biosensor for whole blood creatinine testing. StatSensor Creatinine incorporates patented Multi-Well™ technology. StatSensor Creatinine's advanced technology enables simple, rapid, and accurate assessment of renal function by fingerstick capillary blood sampling at the point of care.
Simple Testing Procedure
- Place biosensor in meter
- Lance finger to create blood drop
- Apply blood drop to biosensor
- Read creatinine results
Benefits
- Virtually painless, 1.2 µL fingerstick capillary blood sample
- Creatinine and eGFR results in 30 seconds
- Cleared for use by point-of-care (POC) personnel
Radiology and cardiac catherization
Minimize risk of contrast induced nephropathy (CIN) – StatSensor Creatinine provides rapid, real-time renal assessment prior to contrast imaging.
Improve productivity and workflow – StatSensor Creatinine POC testing can prevent costly procedure room cancellations and workflow interruptions when patients arrive for imaging appointments without a current creatinine/eGFR workup.
Improve patient satisfaction – StatSensor Creatinine testing can prevent patient dissatisfaction when renal function must be determined before an imaging procedure and has not yet been tested when the patient arrives for the procedure.
Oncology
Reduce medication risks – Renal impairment is frequent in cancer patients, who are at high risk of chemotherapy-induced renal toxicity. StatSensor Creatinine provides important kidney function information for chemotherapy drug selection and dosing.
Improve patient satisfaction – Many cancer outpatients need to have their kidney function assessed before receiving their chemotherapy treatment. Obtaining a venous blood sample and sending it to a laboratory for creatinine/eGFR testing could delay chemotherapy for several hours. A chemotherapy treatment that might take a couple of hours could turn into a full day and cause patient dissatisfaction. With StatSensor Creatinine, a capillary blood sample can be obtained and renal function can be quickly determined.
ED: Improve patient triage, expedite patient care
Various triage patient care protocols in the emergency department may require rapid assessment of renal function in order to triage the patient. In addition, many ED therapies require timely administration of drugs and adjustment of drug dosage based on renal function. StatSensor Creatinine accurately assesses renal function with a simple, 30-second test that can be easily performed in the ED.
01928 704040 | uk-info@novabio.com
Specifications
StatSensorCreatinine Connectivity Meter
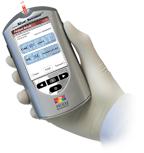
Weight: 220 g (0.8 lb)
Size: 153 mm x 82.5 mm x 46 mm
(6.0 in x 3.25 in x 1.8 in )
Tests:
Tests Measured: Creatinine and Hematocrit
Test Reported: Creatinine and eGFR
Test Time: 30 seconds
Test Strip Volume: 1.2 µL
Test Methodology: Electrochemistry
Selectable eGFR Calculation Equations:
MDRD, IDMS traceable MDRD, Schwartz, Counahan-Barratt and Cockroft-Gault
Sample Types & Operating Modes:
Whole Blood: Arterial, Venous, Capillary
Measurement Range:
Creatinine 27-1056 µmol/L (0.3-12.0 mg/dL)
Operating Ranges:
Temperature: 15°C- 40°C (59°F-104°F)
Altitude: 4,572 m (Up to 15,000 ft)
Humidity: 10% to 90% relative humidity
Data Storage:
Patient Tests: 1,000 tests
QC Tests: 200 tests
Users: 8,000 users
Connectivity:
Data Output Port: Etherent (10 Mbit)
Meter Data Output: RJ-45 Ethernet Port
Protocol: TCP/IP Ethernet 100 Mbit
Standard: POCT1-A Compliant
Setup Program: NovaNet™ Web-Based Instrument Manager Software
Battery Information:
Type: 3.7V Li Polymer Rechargeable Battery
Features: Rechargeable/Replaceable
Life: 8 hours in use (approximately 40 tests
w/barcode scans)/12-24 hours standby
Docking Station:
Optional accessory; desk or wall mount charging station with RJ-45 Ethernet jack. 3 LED indicator lights showing connection status, transmit/receive data, and charging status. Extra battery slot included for recharging and storage of spare battery.
Reagents and Strips:
Strips: 25 strips per vial; sold separately
QC: Three levels (Low, Normal, High)
Linearity: Five levels available
Test Strip & QC Stability:
12 months (4-8°C)
3 months open-vial stability
Additional Features:
- Color touchscreen
- Built-in barcode scanner
- Traditional QC with target values assigned to QC materials
- Numeric or pass/fail QC
- QC prompting or QC lock out
- Abnormal and critical ranges flagged by color highlighting
- Canned or free text user comments
- Reject results at meter (optional) for repeat testing
- Accept patient ID or accession number
- Use of operator password (lockout)
- Entry of ICD-9 diagnosis codes
- Order physicians ID
- Slope and intercept adjustment
- Custom, web-based meter configuration by location
- Connect to hospital ADT
- Connect to all LIS/EMR
- POCT1-A2 data output
- Optional Carrying Case
StatSensor Xpress Creatinine Meter

Xpress Creatinine not available in the U.S. and Canada
Designed for applications that do not need data interfacing and connectivity, Xpress Creatinine retains all of the analytical performance features of StatStrip Creatinine test strips.
Weight: 75 g (0.2 lb)
Size: 91 mm x 58 mm x 23 mm (3.6 in x 2.3 in x 0.9 in)
Data Storage:
Patient & QC Tests: 400 tests total (FIFO)
Connectivity:
Data Transfer: Strip Port Connection to USB
Data Program: Nova Microsoft-Excel based data transfer software
Battery Information:
Type: 3V Li Button Battery
Features: Replaceable
Life: Minimum 600 tests
Additional Features:
- LCD black/white display
- Large numeric display (30 mm)
- Traditional QC with target values assigned to QC materials
- Units of measure based on meter ordered (mg/dl or mmol/L models)
- Automatic shut-off when not in use
- Automatic sample detection and analysis start
- Automatic sample counter with date/time stamp for data tracking
Certifications and Compliance: Nova Biomedical is certified to FDA Quality System Regulations and EN ISO 13485:2016 Complies to IVDD Tested according to: EN 61010-1:2010, EN 61010-2-101:2015, EN 60825-1/A1:2014.
CSA Standards. Patented Technology. Patents 6,287,451 / 6,837,976 / EP 1 212 609 / CA 2,375,092 / EP 1497 449 / JP 405 0078
Specifications current as of revision date.